Amino acid shampoo system
Why emphasize the issue of salt content
1 First of all, we from a variety of amino acid table live molecular structure characteristics, sodium chloride and other inorganic salts on its thickening effect is not ideal, the effect is general, only the salt to thicken the immediate addition of a large amount is also difficult to meet the requirements, and will affect the solubility of amino acid table live;
2 we do amino acid shampoo, to get a good experience, naturally need to add conditioning agents, especially to do silicone oil-free shampoo, to a large extent can not avoid the use of some better conditioning polymer cationic conditioning agent, such as polyquaternium-10, especially in the amino acid system is more sensitive to salt content, easy to produce salt precipitation.
In fact, the main reason for considering the salt content is because of the hard demand for the use of these cations, so the salt content has to be considered.
Salt content
What are the effects on the amino acid system
1 salt content will not only have an impact on the solubility of cationic conditioning agents, too much will produce salt precipitation resulting in turbidity.
2 salt content affects the solubility of amino acid table live, reducing its solubilization effect and further aggravating the cationic salting effect.
3 Excess salt can cause the thickening system to collapse, making the consistency drop rapidly
PH value
Role in amino acid shampoos
Commonly used amino acid table activities are basically strong bases and weak acid salts, which means that their aqueous solutions are basically alkaline in appearance.
Of course, there are exceptions such as sodium cocoyl glutamate, sodium lauroyl glutamate, etc., because their molecular structure contains two carboxyl groups, and only one carboxyl group is neutralized, so it is weakly acidic, while most other amino acid table live aqueous solutions are basically weakly basic.
Let’s take sodium cocoylaminopropionate as an example, its 30% aqueous solution, the PH value is about 8, let’s take a look at the following data
Comparison chart of foam performance test between sodium cocoylaminopropionate and sodium lauroyl alanine
1. Test conditions
1.1 Test temperature 40℃±1℃
1.2 Test liquid volume 100g, corresponding to 0.25% active substance concentration
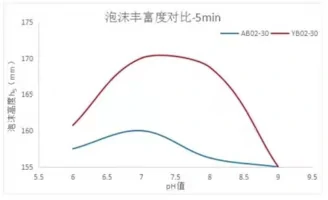
From the graph we can see that sodium cocoylaminopropionate this amino acid table live in the PH value of 7-8 this range value foaming efficiency is the best, in the weak acidic environment is a declining trend, then to make him in the system to show a better foaming effect, it is necessary to adjust the PH value to this range.
From the results of our tests, most of the amino acid table activities basically have this characteristic, which means that in the amino acid system, the PH value plays a great role in influencing the foaming performance of amino acid table activities, which requires us to adjust to a relatively reasonable PH value to ensure the foaming performance of amino acid table activities.
The importance of synergy
1 shampoo system inside, we can be subdivided into table activity system, conditioning system, thickening system, antiseptic system, aroma, efficacy system, concept system, etc., and these systems exist interrelated.
2 As an example, CAB35 is table-active and can act as a foaming agent as well as a thickening agent.
3 between the various systems they will form an interlocking, a raw material can play multiple roles, we need to make good use of this relationship between them, you can achieve twice the result with half the effort.
*Disclaimer: The content contained in this article comes from the Internet, WeChat public numbers and other public channels, and we maintain a neutral attitude toward the views expressed in the article. This article is for reference and exchange only. The copyright of the reproduced manuscript belongs to the original author and the institution, and if there is any infringementPlease contact Jetson Chemical for deletion
