Surfactants are an important class of fine chemicals. When they are dissolved in aqueous solution at a very low concentration or dispersed, they are preferentially adsorbed on the surface or other interfaces of the solution, so that the surface or interfacial free energy (or surface tension) is significantly reduced and the interfacial state of the system is changed.
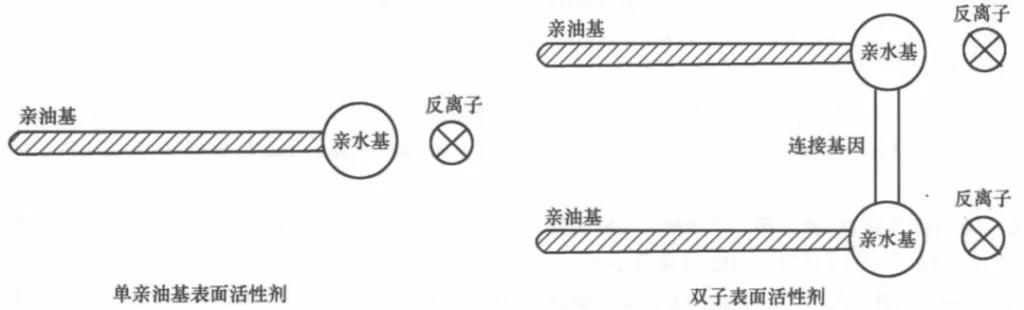
The molecules of surfactants are generally composed of hydrophobic and hydrophilic groups, so they are called amphiphilic compounds.
The molecule has an asymmetric structure, part of which is easily soluble in water and has hydrophilic nature, called hydrophilic group, common groups include carboxyl group, sulfate group, sulfonic acid group, phosphate group and quaternary ammonium group, etc.; the other part is insoluble in water but easily soluble in oil and has lipophilic nature, called lipophilic group, also called hydrophobic group, generally straight chain hydrocarbon (aromatic) containing C8~C18 carbon, such as carbon radon chain, carbon fluorine chain, polysiloxane and polyoxypropylene group Translated with www.DeepL.com/Translator (free version)
Baryonic surfactants and biosurfactants, molecules with at least two or more hydrophilic groups (polar groups), or at least two or more lipophilic groups (hydrocarbon chains, carbon silicon chains or carbon fluorine chains), and linked by chemical bonds at or near the hydrophilic groups, are called baryonic surfactants
*Disclaimer: The content contained in this article comes from the Internet, WeChat public numbers and other public channels, and we maintain a neutral attitude toward the views expressed in the article. This article is for reference and exchange only. The copyright of the reproduced manuscript belongs to the original author and the institution, and if there is any infringementPlease contact Jetson Chemical for deletion
